by Kristen Minogue

Melissa McCormick kneels over a cranefly orchid. (Yini Ma)
The secret is in the soil. In one of the oddest couples in the natural world, orchids need fungi to grow. But finding those fungi can be tricky, until a new study from the Smithsonian Environmental Research Center (SERC) used DNA to find them in more places than anyone suspected.
There are 14 federally endangered or threatened orchids in the U.S. alone, and dozens more are endangered or threatened at the state level. Figuring out how to restore any single species is difficult, because there are so many different kinds.
“You’re talking about the largest plant family in the world,” said Melissa McCormick, lead author and SERC molecular ecologist. Orchids grow from islands off Antarctica to the Arctic Circle and just about everywhere in between. “They grow in darn near every environment on Earth.” But for all their diversity, the planet’s 26,000-plus known orchid species have one thing in common: None can germinate in the wild without a suitable fungus.

Mauve sleekwort (Liparis liliifolia), endangered in three states. Only one species of fungus can make it germinate. (Gary Van Velsir)
Orchids seeds are little more than specks of dust. They have virtually no nutrient stores of their own. Any chance of germinating and blossoming into lady’s slippers or pogonias depends on their finding the right fungus. But for every orchid, the “right fungus” means something different. Some can pair up with five or more different fungi. Others, like mauve sleekwort, match just one.
This posed a dilemma for scientists working to restore endangered and threatened orchids in the wild. They couldn’t plant orchid seeds just anywhere and expect them to grow. Without the right fungi in the soil, orchid seeds would lie dormant, or just die. But how to figure out where those fungi are?
In the 1990s, SERC plant ecologist Dennis Whigham devised a strategy with then-postdoc Hannah Rasmussen.
“One of the things that she and I kept batting around was, how can we work with orchid seeds in nature, because they’re so teeny?” Whigham said. They came up with a technique to put orchid seeds in mesh packets and bury them in the soil. The mesh allowed just enough space for fungi, while keeping the orchid seeds from getting accidentally swallowed by hungry worms or other creatures chewing on the soil. If the seeds germinated, the right fungi were nearby.
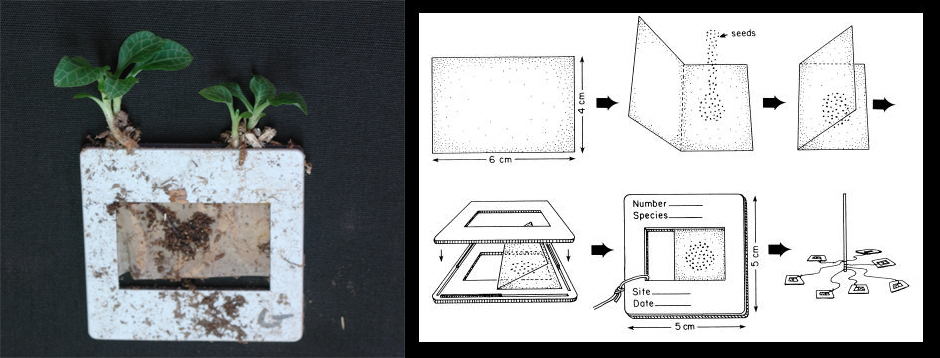
Left: Downy rattlesnake plantain orchids sprout from a seed packet. Right: After placing orchid seeds inside the mesh packet, scientists buried them at various distances around an orchid in the field. (SERC)
For two decades, Whigham and Rasmussen’s technique was the modus operandi for finding hot spots of potential orchid growth. But it led some ecologists to assume the ideal fungi only existed in spots where orchid seeds germinated. That assumption was wrong.
More recently, a string of molecular ecologists began using DNA to search for fungi in the soil, with a technique called next-generation sequencing. Next-generation sequencing uses DNA primers— tiny fragments of DNA designed to latch onto larger chunks of DNA and create millions upon millions of copies. Because the primers are very general, they’re supposed to give a picture of just about everything in an environment.
“The idea is that it ought to pick up everybody,” McCormick said. “In reality, it doesn’t pick up everybody. Some things it picks up better than others. And it turns out that some of these orchid fungi it doesn’t pick up very well.”

Downy rattlesnake plantain (Goodyera pubescens) in full bloom. (Melissa McCormick/SERC.)
In the new study, their University of Mexico colleague Lee Taylor designed new primers to pick up what next-generation sequencing had missed. Taylor’s primers latched onto only the DNA of the fungi for three orchids: downy rattlesnake plantain (Goodyera pubescens), mauve sleekwort (Liparis liliifolia) and cranefly orchid (Tipularia discolor). Then, the team set out to compare the DNA findings with Whigham’s seed packet technique. The team buried hundreds of mesh packets with orchid seeds throughout the SERC forest, checking every few months to see if any orchid seeds had germinated. After two years, they collected all the packets while McCormick took DNA soil samples from where they had been buried.
Their discoveries, published March in Journal of Ecology, revealed that orchid-friendly fungi were far more widespread than anyone had realized. McCormick’s DNA primers picked up generous amounts of fungi almost everywhere orchid seeds germinated. But they also detected the fungi in places where orchids did not germinate. Fungi appeared in sparser amounts in anywhere from 25 to 50 percent of the sites without germination, depending on the orchid.

The cranefly orchid (Tipularia discolor), which puts forth a single speckled leaf in winter (Melissa McCormick/SERC)
For orchids to grow, this means it’s not enough for the right fungi simply to be present. They need to be abundant.
With DNA, scientists could detect fungi that would otherwise have remained invisible. But it would be a mistake to think DNA has all the answers. “If you detect fungus in the soil using just molecular techniques, that does not tell you whether or not orchids will germinate there,” McCormick said. On the other hand, “if you only look at seed germination, that doesn’t tell you everywhere the fungus is.”
While many of the hot spots DNA uncovered don’t have enough fungi for orchids to grow yet, with the right help that could change. In an earlier experiment, McCormick and Whigham added different items to the soil to see what boosted the fungi. “Adding wood made the fungi really much happier,” she said, at least for the fungi those particular three orchids need.
Now, thanks to DNA, scientists can pinpoint more spots where orchid-friendly fungi could benefit from reinforcements. Mother Nature’s odd couple has a fighting chance.

